02
AI Snaps
.svg)
.svg)
01
Our Work

.svg)
03
About Us
.svg)
05
Contact Us
.svg)
06
Client Success

.svg)
07
Blogs
.svg)
08
Careers
.svg)
Book A Call
Need Help In Building Your Brand?
Click the button below & book a call with our founder directly.

Rishabh Jain
Managing Director
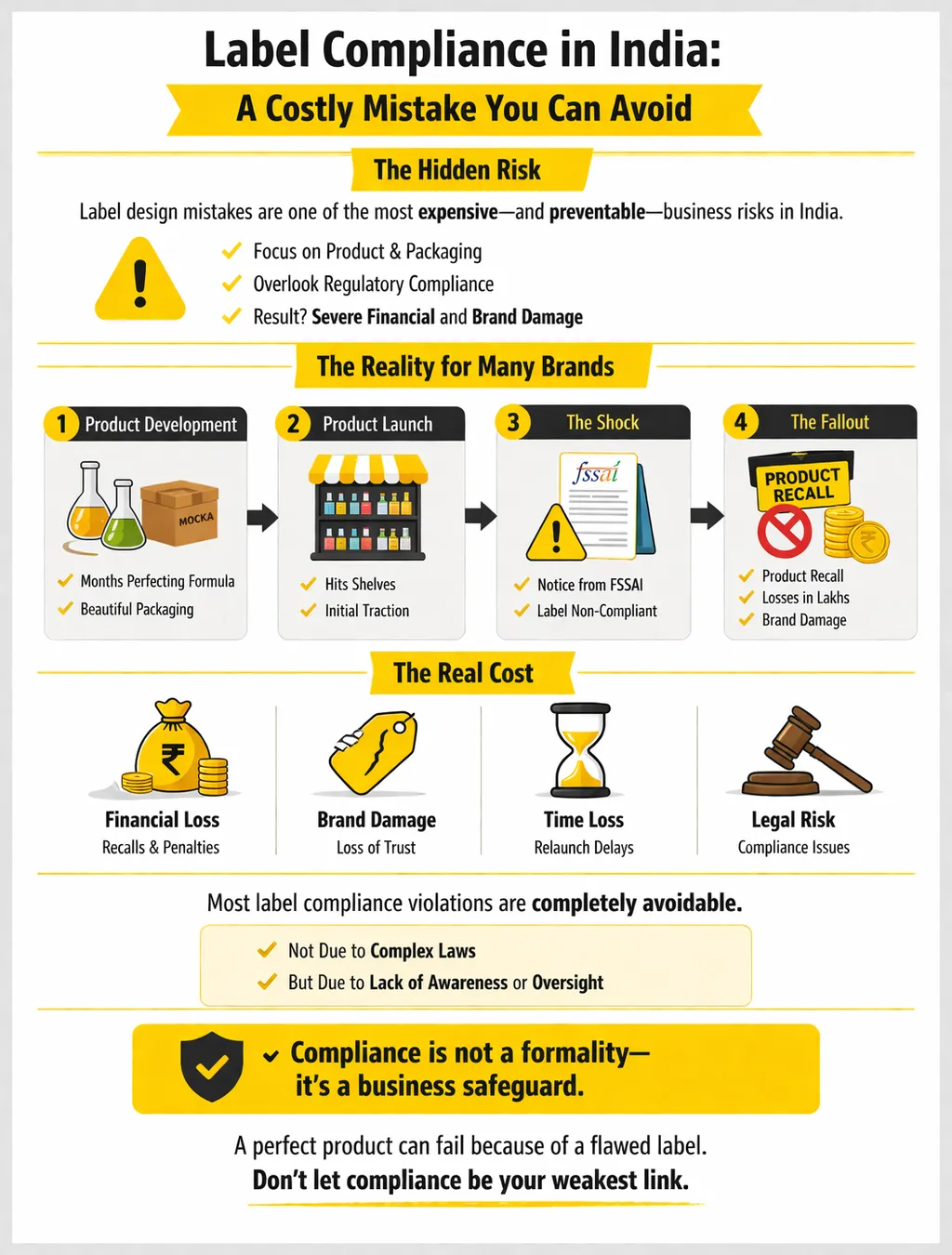
Label design compliance issues in India are responsible for some of the most painful and avoidable business setbacks brands experience every year.
You spend months perfecting a product. The formula is right, the packaging looks great and you're finally ready to launch.
Then, weeks after hitting shelves, a notice arrives from FSSAI. Your label is non-compliant. The product gets pulled. The recall costs you lakhs.
The brand takes a hit it may never fully recover from. Most of these violations were entirely avoidable.
When we talk about label design compliance issues in India, we're talking about the gap between what a label shows and what the law requires it to show.
Every product sold in India must carry certain mandatory information, and that information must be presented in a specific way. The font can't be too small.
The language may need to be in Hindi. The claims you make have to be defensible. There are two categories of label elements worth understanding:
Mandatory elements are non-negotiable. These are things like the product name, MRP, net quantity, manufacturer's address, batch number, and expiry date.
Leave one out, and you're already non-compliant.
Voluntary elements are claims you choose to make, things like "organic," "100% natural," "dermatologically tested," or "sugar-free."
These aren't required, but once you put them on the label, they become legally scrutinised. You need proof to back them up.
Most label compliance issues in India arise in one of three ways, a mandatory element is missing, a voluntary claim can't be substantiated or the formatting doesn't meet the required standards. Often, it's a combination of all three.
Let's be direct about the stakes. Non-compliance with Indian labeling laws isn't just a slap on the wrist.
Depending on the violation and which regulatory body catches it, the consequences can include:
And those are just the legal consequences. The reputational fallout of customers losing trust, retailers dropping your product, and media coverage of a recall can be far harder to recover from than any fine.
For businesses looking to export, the stakes go even higher. International markets have their own labeling requirements and a product that's non-compliant in India often fails import checks in other countries too.
One reason label compliance is so complicated in India is that no single authority governs everything. Different products fall under different regulators, and sometimes multiple regulators apply to the same product.
Here's who you need to know:
If you make or sell food, the Food Safety and Standards Authority of India (FSSAI) is your primary regulator. It defines what information must appear on food labels.
This includes nutritional declarations, allergen disclosures, and health claims. FSSAI also sets rules for how your license number must be displayed on packaging.
If you don’t follow these regulations, you risk non-compliance. In fact, FSSAI violations are one of the most common reasons for food product recalls in India.
The Legal Metrology Division focuses on how you communicate price and quantity. It does not depend on what your product is. It applies to how you label and sell it.
MRP declarations are covered under this law. Net weight or volume must also be clearly mentioned. Manufacturer and packer details are required as well. These rules apply to almost every packaged product sold in India.
The Central Drugs Standard Control Organisation regulates drugs, medicines and medical devices. If your product falls in this category, labeling rules become much stricter.
You must include warning labels where applicable. Dosage instructions are mandatory. Storage conditions must be clearly defined.
Batch traceability is also required. These are among the most tightly enforced labeling standards in India.
BIS certification is mandatory for a growing list of products in India. If your product falls under a BIS category, the certification mark must appear on the label in a specific way.
For herbal, Ayurvedic, Unani, Siddha, and homeopathic products, AYUSH sets the labeling framework. This is a fast-growing category with evolving regulations and many brands in this space are still operating on outdated label formats.
The challenge is that these regulators don't always coordinate well. A product like a herbal supplement could technically fall under FSSAI, AYUSH, and Legal Metrology simultaneously.
Understanding which rules apply and where they overlap takes real expertise.
Behind the regulatory bodies are the actual laws. These are the legal frameworks that give regulators their authority:
The Food Safety and Standards Act, 2006 is the cornerstone of food labeling in India. It defines everything from how ingredients must be listed (in descending order of weight) to what constitutes a misleading health claim.
The Legal Metrology Act, 2009 governs all pre-packaged commodities. It sets rules for how MRP must be declared, how net quantity is measured and displayed, and what manufacturer information is required.
The Drugs and Cosmetics Act, 1940, despite its age, remains the governing law for pharmaceutical and cosmetic labeling. It has been amended numerous times since independence, but the core framework still stands.
The Consumer Protection Act gives consumers the right to accurate information. Labels that mislead, even unintentionally, can trigger consumer complaints and legal action under this law.
BIS Certification Rules apply to product categories that require Bureau of Indian Standards certification. These rules specify exactly how the ISI mark must be displayed, and misuse of the mark is a serious offence.
The complexity increases because these laws often overlap. A single label on a packaged food product might need to satisfy FSSAI rules, Legal Metrology requirements, and Consumer Protection Act standards all at the same time.
In India, you don’t lose your product because it’s bad. You lose it because your label wasn’t taken seriously. Compliance isn’t paperwork, it’s survival.
If your label can’t stand up to a regulator’s scrutiny, it won’t stand up to the market either. Compliance is not a constraint—it’s a credibility signal.
— Rishabh Jain, Founder, Confetti Design Studio
So what actually needs to be on your label? While requirements vary by product category, here's what most packaged products sold in India must include:
Beyond what to include, the law also governs how it's displayed. Minimum font sizes are specified for different label elements.
Certain information must appear on the principal display panel. Nutritional information must follow a prescribed format. These formatting rules catch many brands off guard.
After working with brands across food, pharma and personal care, certain mistakes come up again and again. Here are the ones most likely to get you into trouble:
This is probably the single most common issue. Indian labeling laws specify minimum font sizes for elements like MRP, net quantity, and manufacturer details.
When brands try to fit a lot of information into a small label, text gets compressed and you're suddenly non-compliant.
The fix sounds simple, but it requires rethinking the whole label layout.
Some brands skip manufacturer addresses because they think the brand name is enough.
Others omit customer care details because they don't think anyone reads them. Regulators do. Every mandatory field must be present, complete, and accurate.
"100% natural," "clinically proven," "zero side effects" - these phrases appear on thousands of Indian products. But without documented proof, they're legally indefensible.
FSSAI and the Consumer Protection Act both specifically prohibit claims that mislead consumers, and "natural" is one of the most scrutinised terms in food labeling.
Getting the MRP wrong, whether it's calculated incorrectly, printed in the wrong format, or doesn't include all taxes, is a Legal Metrology violation.
It seems like a simple thing, but MRP errors are surprisingly common, especially when products are relaunched or prices are revised.
India's labeling rules require that key information appears in English and Hindi on most products. Some categories require regional languages as well.
Many brands, especially those entering India from other markets, produce English-only labels that are immediately non-compliant.
Using the FSSAI logo incorrectly, displaying a BIS mark on an uncertified product, or showing an outdated licence number are all serious violations.
Regulatory logos and marks come with strict usage guidelines that must be followed exactly.

Beyond the standard mandatory requirements, food labels in India must declare allergens (peanuts, tree nuts, gluten, dairy, etc.) clearly.
Nutritional values must be provided per 100g or per 100ml and per serving. FSSAI license numbers must be prominently displayed.
And any health or nutrition claim, from "high in protein" to "low fat," must meet specific thresholds defined in the regulations.
Drug labeling in India is among the most tightly regulated in Asia. Warning labels, contraindications, storage conditions, and batch traceability are all mandatory.
Missing a single warning label on a pharmaceutical product isn't a minor oversight. It's a patient safety issue and a CDSCO enforcement trigger.
Cosmetic labeling must list every ingredient using INCI (International Nomenclature of Cosmetic Ingredients) names. Claims like "whitening," "anti-ageing," or "SPF" are closely scrutinised.
Products that fall between cosmetics and drugs, often called "cosmeceuticals," face particular ambiguity, as they may need to comply with both the Drugs and Cosmetics Act and cosmetic labeling standards.
This is an often-overlooked area. FSSAI and the Legal Metrology Department have both issued guidelines requiring that product information displayed online must match the physical label.
Brands that list products with incomplete or incorrect online information, even if the physical label is correct, are exposed to compliance risk.
India's linguistic diversity creates real labeling challenges. The general rule for most packaged goods is:
For exporters selling into India, this means labels designed for Western markets almost never meet Indian requirements out of the box. A complete relabeling exercise is usually necessary.
For Indian brands exporting abroad, the situation is reversed. Indian labels typically need to be supplemented with destination-country languages, and sometimes the format requirements conflict with Indian standards.
Many brands conflate packaging compliance with label compliance. They are related, but they are different.
Packaging refers to the physical container, such as the bottle, box, pouch, or bag. Packaging materials, especially for food, have their own compliance requirements related to food safety and material standards.
Labeling refers to the information printed on or attached to that packaging. This is where the regulatory detail lives.
Both are regulated, and both can create compliance problems independently. A product can have perfectly compliant packaging and a non-compliant label, or vice versa.
Understanding where one ends and the other begins is important when auditing your compliance.
To make the consequences concrete:
Under the Food Safety and Standards Act, food businesses can face penalties of up to Rs. 3 lakh for misleading advertisements and labeling, with higher penalties for repeated violations or violations that cause harm.
Under the Legal Metrology Act, penalties for incorrect declarations can range from Rs. 2,000 to Rs. 25,000 for a first offence, with repeat offences carrying imprisonment.
Under the Drugs and Cosmetics Act, penalties for labeling violations involving drugs can include imprisonment of up to three years.
Beyond individual fines, FSSAI has the authority to suspend or cancel food business licences for persistent non-compliance, which can be an existential threat for a food brand.
The brands that avoid label compliance issues in India generally do three things consistently:
1. Build compliance into the design process from day one. The most expensive label mistake you can make is one you discover after printing 50,000 units. Compliance should be verified before the design is finalised, not after.
2. Conduct regular compliance audits. Regulations change. FSSAI updates its labeling standards. Legal Metrology revises formats.
A label that was compliant three years ago may not be compliant today. Periodic audits, at least annually, catch these changes before they become violations.
3. Work with people who understand both design and regulation. This is where many brands fall short. A designer who doesn't understand labeling law will produce a beautiful label that fails a compliance check.
A compliance consultant who doesn't understand design will produce a label that meets regulations but communicates poorly. The sweet spot is teams that genuinely understand both.
Pre-launch checklist to follow every time:
India's labeling landscape is changing faster than it has in decades. A few trends worth watching:
QR codes for traceability. FSSAI has already begun pushing for QR codes on food labels that link to detailed product information including batch records, sourcing details, and nutritional data.
This is likely to become mandatory in more categories over the next few years.
Smart labels and digital integration. The boundary between physical labels and digital product information is blurring.
Brands that build digital infrastructure now will be better positioned when regulations require it.
Sustainability disclosures. Environmental claims like "recyclable," "compostable," and "carbon neutral" are under increasing scrutiny globally, and India is likely to follow with formal guidelines.
Brands making these claims today should be building the substantiation records to defend them.
AI-powered compliance checking. Several platforms are beginning to use AI to cross-reference label designs against regulatory databases in real time.
This technology is still maturing, but it's likely to change how compliance review works for high-volume brands.
Label compliance in India isn't glamorous work. It's detailed, sometimes tedious and often requires navigating rules that feel like they were written to contradict each other. But it's fundamental.
Every product you put on the Indian market carries your brand's name. A non-compliant label doesn't just create legal risk.
It signals to regulators, retailers, and customers that you haven't done the work. Getting it right signals the opposite.
The good news is that for brands willing to invest in compliance from the start, the risks are very manageable. The rules, once understood, are workable.
And the brands that build compliance into their process rather than treating it as an afterthought end up with stronger products, fewer delays, and a lot less stress.
Need help designing labels that meet Indian regulatory requirements without sacrificing visual quality? Confetti Design Studio specialises in compliance-first label design for food, pharma, cosmetics, and consumer goods. Get in touch before your next print run.
What are the most common label design compliance issues in India? The most frequent problems are missing mandatory information (MRP, manufacturer details, batch numbers), incorrect font sizes, unsubstantiated marketing claims, and language violations. These issues affect brands across every category.
Which authority regulates product labeling in India? There's no single authority. FSSAI regulates food labeling, the Legal Metrology Department governs quantity and pricing declarations, CDSCO handles pharmaceutical labels, BIS certifies product standards, and AYUSH covers herbal and Ayurvedic products. Many products fall under multiple regulators simultaneously.
Is Hindi mandatory on product labels in India? For most categories of packaged goods, yes, particularly food products. English is always required, and Hindi is required in most consumer product categories. Regional languages may also be required depending on the category and state.
What happens if a product label is non-compliant in India? Consequences range from fines (potentially in lakhs) to product seizures, licence suspension, forced recalls, and in serious cases, criminal prosecution. The reputational damage from a public recall often exceeds the direct financial penalties.
How often should brands audit their labels for compliance? At minimum, once a year, and immediately whenever regulations change or a product is reformulated, repriced, or repackaged. Given how frequently Indian labeling standards are updated, annual audits are the bare minimum for brands with significant market presence.


.svg)